NCKU Researchers Achieve New Milestone in RNA Biology, Opening a New Chapter in Hypoxia Biology
A research team at National Cheng Kung University (NCKU) has uncovered a new mechanism by which the hypoxia-inducible factor-1α (HIF-1α) rewires RNA metabolic pathways in the nucleus, revealing a previously unrecognized strategy used by cancer cells to adapt to hypoxia. The study, led by Dr. Pai-Sheng Chen from the Department of Medical Laboratory Science and Biotechnology at NCKU, was published in the international journal Nucleic Acids Research. The new research reveals that HIF-1α, aside from serving as a transcription factor, can "rewire" RNA metabolic pathways within the nucleus. A non-canonical role for HIF-1α: redirecting DGCR8 to the RNA exosome for snoRNA degradation and translational modulation. (Nucleic Acids Research, 2026). doi: doi.org/10.1093/nar/gkag070.
Important discoveries in RNA biology, including work related to microRNAs (miRNAs), have received major international recognition in recent years, allowing scientists to further uncover the mysteries of the molecular world
within cells. Following their 2024 publication in the international journal Nucleic Acids Research, the research team led by Dr. Pai-Sheng Chen has now published their latest findings in the same journal. This process affects ribosomal RNA modification and protein production efficiency, providing a novel perspective on how tumors adapt to hypoxia and growth stimuli.
The human body is composed of tens of trillions of cells, and each cell contains thousands of different types of molecules. Just as cells need to interact with each other, molecules within cells must maintain real-time dynamic communication to sustain the cell's survival advantage. For a cell, hypoxia is both a stress and a stimulus. Particularly during the growth of solid tumors, once cancer cells adapt to the hypoxic environment and overcome this critical survival bottleneck, the progression of the disease often becomes difficult to control.
The first author Jie-Ning Li stated that the survival pressure under hypoxic conditions forces cancer cells to evolve a complex and delicate adaptation mechanism. First, hypoxia-inducible factor-1α (HIF-1α) accumulates dramatically and activates hundreds to thousands of downstream target genes, thereby remodeling pathways for cell metabolism, survival, and angiogenesis. Concurrently, HIF-1α blocks the biogenesis of miRNAs, allowing post-transcriptional gene products (mRNAs) to accumulate rapidly without suppression. At first glance, it might seem that the cell's "protein factory" should kick into high gear and begin mass-producing proteins. However, the research team discovered that HIF-1α simultaneously engages another regulatory mechanism. Through the reassembly of the RNA exosome complex, it degrades snoRNAs, which are essential components of the protein factory. This ultimately leads to the suppression of the overall protein production line (translation). Does this phenomenon seem contradictory?
Dr. Pai-Sheng Chen stated that this is not strange at all; rather, it is an extremely efficient protective mechanism employed by cancer cells. Compared to mRNA production (transcription), protein production (translation) consumes an immense amount of energy. If the cell were to indiscriminately initiate the massive production of all intracellular proteins at this time, it would need to expend several times more energy than under normal conditions. Since hypoxic environments are typically accompanied by nutrient deprivation, energy becomes incredibly precious under these circumstances.
If hypoxia is viewed as an emergency situation, the slowdown of overall protein production is like vehicles on a highway universally decelerating and yielding the right of way. Meanwhile, the "ambulances" responding to the emergency, carrying critical hypoxia-induced genes, bypass the traffic through the emergency lane to deliver precisely the proteins needed to cope with the lack of oxygen. Through this mechanism, cancer cells manage to survive in adverse conditions using the most efficient method possible. It turns out that cancer cells are also well-versed in the strategy from Sun Tzu's The Art of War: "Those skilled in defense hide in the deepest depths of the earth," utilizing extreme resource management to survive the hypoxia crisis.
Dr. Chen's team has published a series of breakthrough research achievements. These include the 2018 discovery that HIF-1α non-transcriptionally inhibits Dicer to promote cancer metastasis (Journal of Clinical Investigation, 2018), and the 2024 finding that HIF-1α interferes with Microprocessor assembly to affect miRNA maturation in the nucleus (Nucleic Acids Research, 2024). Their latest breakthrough further decodes how HIF-1α "redirects" DGCR8 to the RNA exosome. Through the snoRNA-rRNA-protein translation axis, this assists cells in regulating energy expenditure under hypoxic conditions, providing critical scientific answers to how tumors adapt to environmental stress (Nucleic Acids Research, 2026).
As for whether there will be a sequel, Dr. Chen smiled and said, "Research has no endpoint, and there are still many unsolved mysteries in this precise cellular regulatory mechanism. But facing the unknown is a kind of enjoyment. However, without the injection of research resources, any scientific dream can only remain a dream, and any difficulty will only become an insurmountable threshold." Therefore, the research team expresses special gratitude for the long-term support from the National Science and Technology Council (NSTC). They also thank NCKU, the Department of Medical Laboratory Science and Biotechnology, the Research Center for Medical Laboratory Biotechnology, the Bioimaging Core Facility of the National Core Facility for Biopharmaceuticals, Academia Sinica, National Taiwan University, and Baylor College of Medicine for their strong support through shared core facilities and research resources, enabling Taiwan’s leading research achievements to continue gaining international recognition.
NCKU stated that Nucleic Acids Research is published by Oxford University Press and is a leading international journal in the field of nucleic acid research. This research provides an important scientific foundation for the future development of biomedicine, particularly in the understanding of disease mechanisms and the development of targeted drugs.
Previous Studies in this Series:
2018 study:HIF-1α promotes autophagic proteolysis of Dicer and enhances tumor metastasis (Journal of Clinical Investigation, 2018). doi: 10.1172/JCI89212.
2024 study:A transcription-independent role for HIF-1α in modulating microprocessor assembly (Nucleic Acids Research, 2024).
Provider: NCKU News Center
Date: 2026-03-17

The research team led by Dr. Pai-Sheng Chen of the Department of Medical Laboratory Science and Biotechnology at NCKU has once again published their nucleic acid research in the top international journal Nucleic Acids Research.
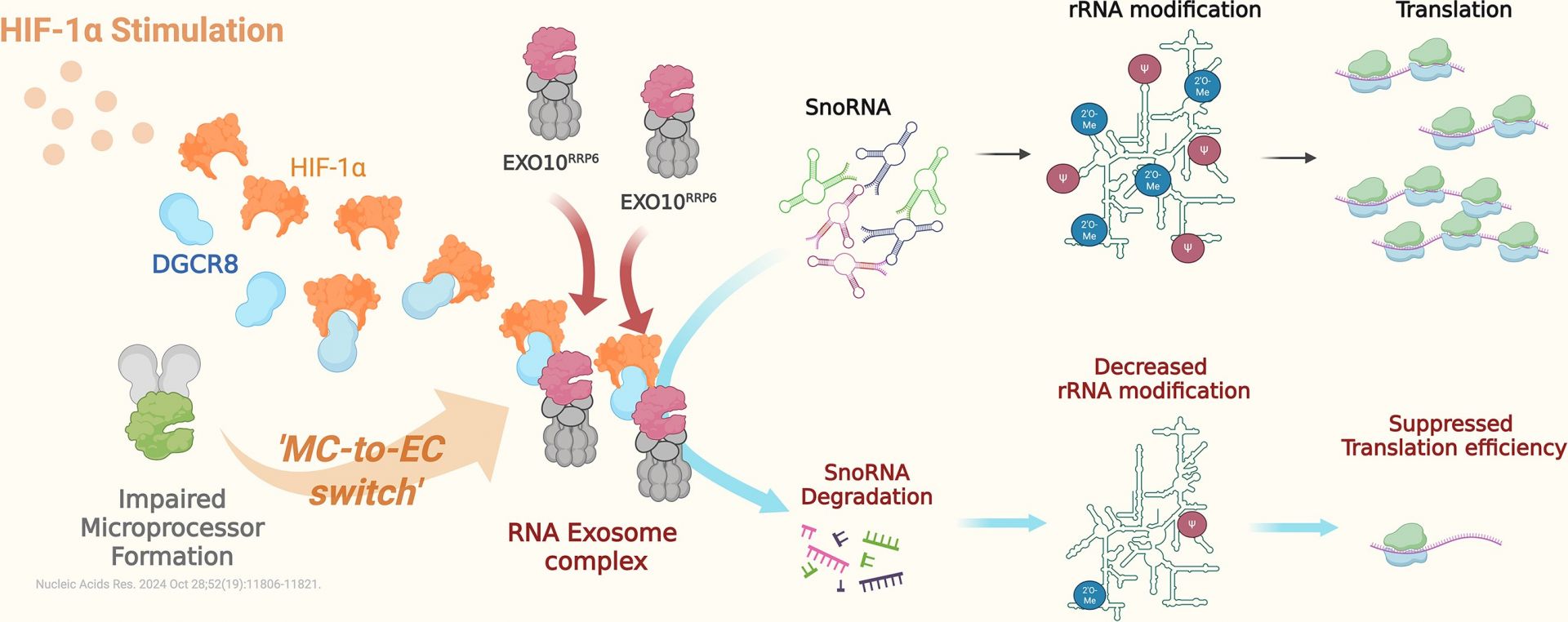
The latest research from Dr. Pai-Sheng Chen's team at NCKU reveals that the hypoxia-inducible factor-1α (HIF-1α) not only acts as a transcription factor but also "rewires" RNA metabolic pathways in the nucleus. This affects ribosomal RNA modification and protein production efficiency, providing a new perspective on tumor adaptation strategies under hypoxia and growth stimuli. The findings have been published in the top international journal Nucleic Acids Research.

The research team led by Dr. Pai-Sheng Chen (first from the left) of the Department of Medical Laboratory Science and Biotechnology at NCKU has once again published their nucleic acid research in the top international journal Nucleic Acids Research. The second from the right is the first author, postdoctoral researcher Jie-Ning Li.

SDG3President Lai Attends Groundbreaking of NCKU Shalun Hospital, Advancing Taiwan’s National Healthcare Development
View more
SDG3NCKU Medical School's Laboratory Animal Center Receives Full AAALAC Accreditation Again
View more



















